
Of note was that history of prior stroke/transient ischemic attack was higher in the no-diabetes group than in the diabetes group (33% vs. There were fewer female patients with diabetes than without (37% vs. Most of the patients with diabetes did not require insulin (30%), while 6% did. In all, 7,624 of the 21,105 patients in the ENGAGE AF-TIMI 48 trial had diabetes, for a rate of 36%. Key secondary endpoints included in the subanalysis were cardiovascular death, stroke/SEE, major adverse cardiovascular events (MACE, a composite of myocardial infarction, stroke, SEE, or death because of cardiovascular cause or bleeding), and all-cause death. The primary endpoint and the primary safety endpoint of interest were the same as in the main ENGAGE AF-TIMI 48 trial. Next, they analyzed edoxaban concentration, anti–factor Xa activity, and international normalized ratio data and compared outcomes of high-dose edoxaban with those of warfarin. The investigators further stratified patients into insulin-treated and non–insulin treated groups and used multivariate Cox regression models to adjust for baseline characteristics across the groups stratified by diabetes status. The presence or absence of diabetes was determined by the local investigator at randomization. Plitt and colleagues set out to further evaluate outcomes of patients enrolled in the ENGAGE AF-TIMI 48 trial, excluding those who were in the low-dose edoxaban group. is ‘shameful,’ say expertsįor the current subanalysis, Dr. “The lower-dose regimen was not approved because there was less protection from ischemic stroke, compared with warfarin.”Įxtent of insulin rationing in the U.S. “Therefore, the higher dose of edoxaban was approved globally for treating patients with AFib,” Dr. 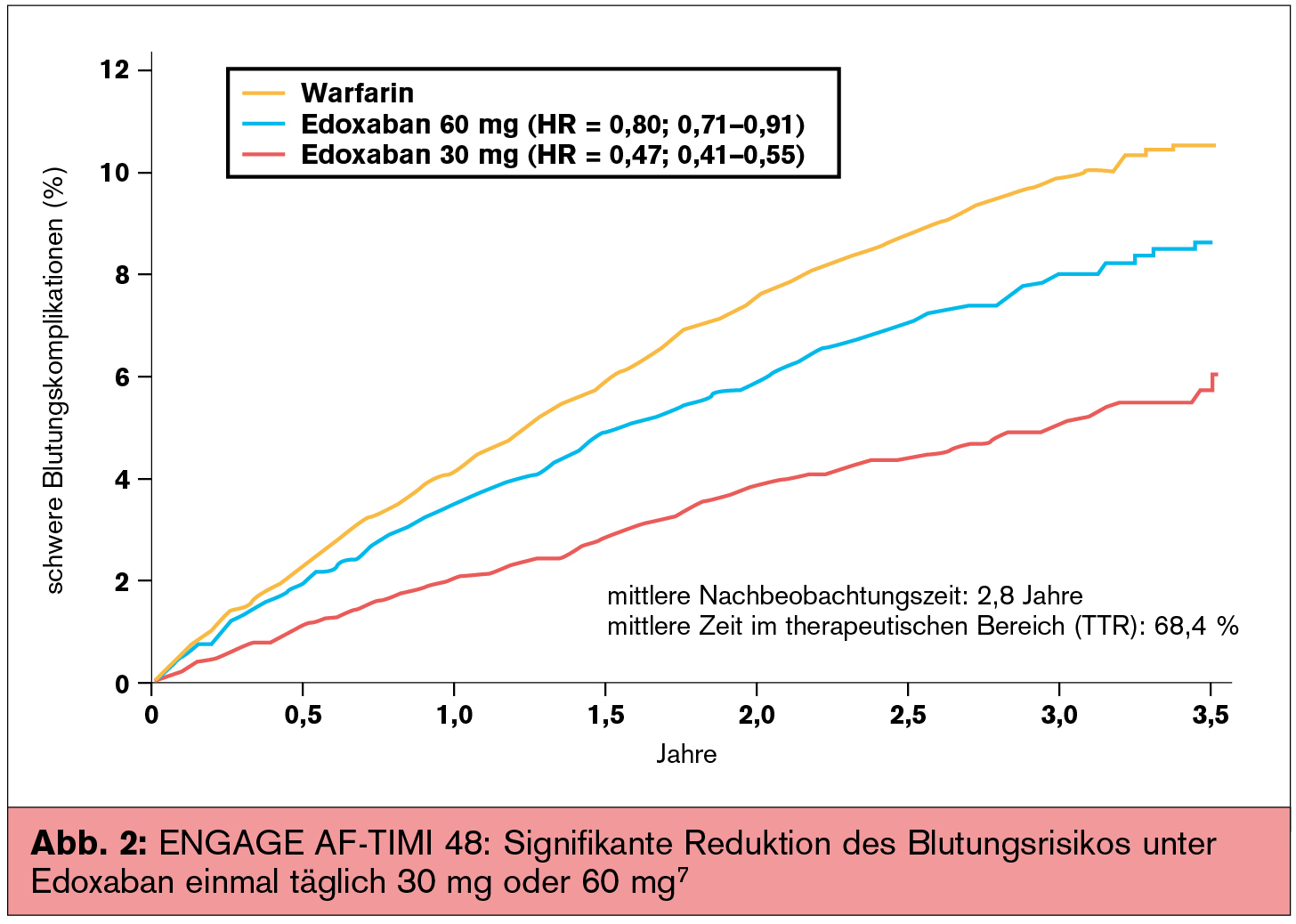
It also significantly reduced major bleeding, cardiovascular death, and net outcomes. The findings showed that edoxaban was noninferior to warfarin in preventing stroke/SEEs. The primary safety endpoint was major bleeding, as defined by the International Society on Thrombosis and Haemostasis criteria. The median follow-up was 2.8 years, and the primary efficacy endpoint was stroke or systemic embolic events (SEEs). The edoxaban dose was reduced by 50% if creatinine clearance reached 30-50 mL/min, patient weight reached 60 kg or less, or there was concomitant use of a P-glycoprotein inhibitor ( N Engl J Med. In the ENGAGE AF-TIMI 48 trial, 21,105 patients with documented AFib within the previous 12 months were randomized to standard-care warfarin or high-dose edoxaban (60 mg daily) or low-dose edoxaban (30 mg daily). “Recommendations for anticoagulation managements vary based on the presence of additional risk factors and which guidelines are followed.” “Given the complex clinical interactions between AFib and type 2 diabetes, care for these patients remains challenging,” she said. Plitt, a cardiology fellow at Mount Sinai Hospital, New York, noted that, although type 2 diabetes is characterized by a prothrombotic and inflammatory state, the mechanism of action by which hyperglycemia and/or insulin resistance leads to the development of atrial fibrillation (AFib) remains unknown. 
The coexistence of and type 2 diabetes further increases thromboembolic risk.”ĭr. “Type 2 diabetes is associated with a twofold increased risk of stroke, and longer duration of diabetes is associated with even higher ischemic event rates. “We know that atrial fibrillation is associated with a fivefold increased risk of stroke,” Anna Plitt, MD, said at the World Congress on Insulin Resistance, Diabetes & Cardiovascular Disease. In addition, the pharmacokinetic and pharmacodynamic profile of the study drug, edoxaban – a novel oral anticoagulant drug and a direct factor Xa inhibitor – was generally similar in patients with and without diabetes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
